MESA Medizintechnik GmbH

Das Qualitätsprogramm für die Medizintechnik
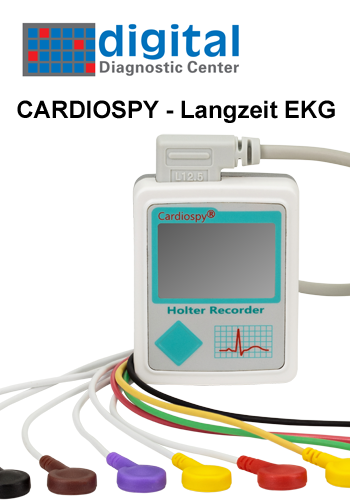
CARDIAX® – das innovative PC-EKG

Der schnelle Kontakt:
Beratung: 08857-6918-0
Fax: 08857-6918-29
email: info@mesamed.de
Der schnelle Kontakt:
Beratung: 08857-6918-0
Fax: 08857-6918-29
email: info@mesamed.de